From semiconductors in our electronics to imaging methods and drug delivery inside the body, there are a vast range of ways we can put the extraordinary properties of 2D materials to use. These traits can include high conductivity, incredible strength, and low weight, among others – all of which have significant potential in developing new industrial materials. But what makes 2D materials special, and how can we make them?
A 2D crystal consists of a single layer of atoms, bonded together at their sides but with nothing above or below them. This changes some of the normal atomic and electronic interactions we would expect to see in an analogous 3D crystal and causes the unique properties we observe. One common example of this is graphene, a single layer of a carbon structure known as graphite. Graphene is extremely strong and has special properties relating to how it conducts heat and electricity that make it distinct from 3D graphite. These properties make it desirable for developing aerospace materials and electronics.
When it comes to making 2D materials there are two options – they can be grown from scratch on top of a substrate, or they can be sliced off larger samples. To grow them, we need a certain ‘recipe’ to start with. This can be difficult to develop, depending on the substrate that we want to use, because atomic interactions with the substrate are a crucial part of the crystal growth process. Also, the growth recipe needs to be optimized to grow only a uniform single atomic layer, without undesired local regions of 3D crystal growth. This can be challenging to achieve.
Cutting samples from existing structures can give us nice 2D layers, but the problem scientists encounter when they cut and study 2D materials is that many of these types of crystals are very prone to becoming contaminated. As they are so thin, dust and tiny particles in the air suddenly become significant in comparison to the tiny atomic layer we are looking at. Some 2D materials are very reactive with air and water, changing their composition rapidly and stopping us from studying the original material. What we need is a way to substantially reduce these contamination risks – which is exactly what CLF scientist Dr. Charlotte Sanders has helped to develop.
Charlotte runs Artemis’ material science program here at the Central Laser Facility, together with fellow researcher Dr. Yu Zhang. The program is designed to provide the scientific community with the tools to study the electronic properties of materials after they are irradiated by a very short, intense laser pulse. Artemis can achieve this experimentation at very short time scales for maximum precision. Charlotte’s field of study focuses on solid-state crystalline materials, including both creating these crystals and studying their electronic properties. She uses the Artemis facility for her research, shining laser beams onto crystals to explore how the electrons in the structure interact with the light.
In this investigation of 2D materials, however, Charlotte played a crucial role as part of a small team working at the ASTRID2 Synchrotron Light Source in Denmark, uncovering the best practices for developing high-quality 2D materials. Previously, the samples would be cut inside a glovebox, and then studied inside a vacuum chamber. Vacuum chambers are almost completely void of atoms, thus reducing contamination risk, and gloveboxes are filled with unreactive gases such as nitrogen or argon for a similar effect. However, transferring the sample from where it is cut to the chamber exposes it to particles in the air. In order to eliminate this undesirable journey, Charlotte and the team have developed a method of slicing 2D material samples inside a vacuum.
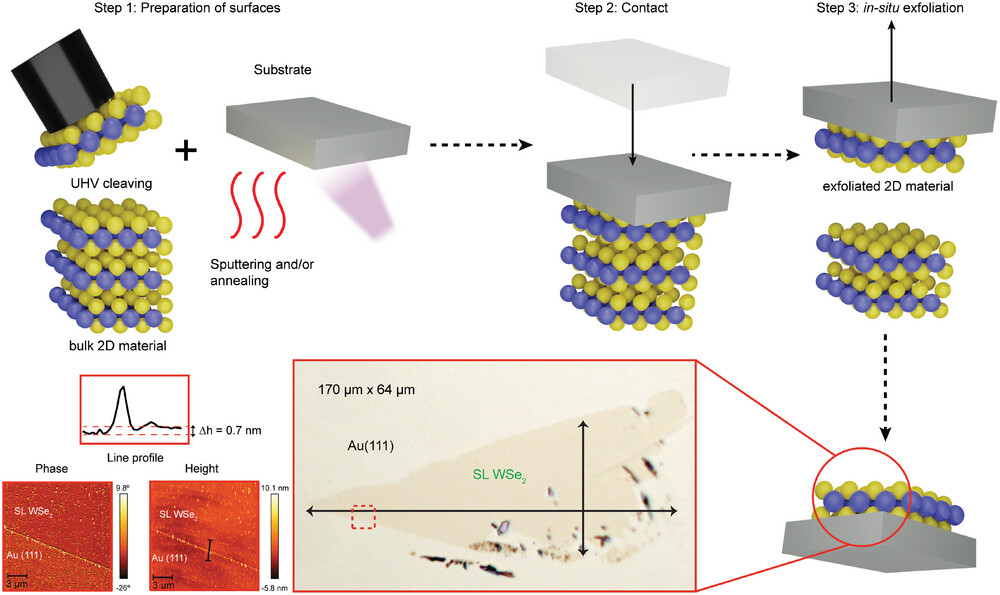
The method works by contacting the 3D source material to the substrate in vacuum. This substrate is chosen such that it reacts with the material just enough to pull off one single layer of atoms, isolating it from the rest of the 3D source. This reaction can be electromagnetic, where the forces of attraction pull away the atoms, or chemical, where a new structure is formed as the atoms interact and bond with one another. This separates a 2D material layer from the larger crystal with minimal outside interaction, allowing it to be generated in a vacuum and removing the need to transfer samples between environments. This method will therefore let scientists like Charlotte derive much purer samples than they have been able to study before, helping to unlock a better understanding of 2D material behaviour. In fact, this method can now be used at Artemis, letting the CLF team study materials that previously we weren’t yet able to! This could help our users develop electronic components, detectors, and medical devices, amongst many other things.
