Their results, published in the prestigious nature journal 'Scientific Reports, identify a novel role for the RTN20 protein and even pave the way for future research relating plant reticulon structure to function.
The Endoplasmic Reticulum (ER) is a multifunctional organelle responsible for a whole host of vital processes that take place within eukaryotic cells. It synthesises and processes proteins and lipids as well as transporting them out of the cell. The basic units of the ER are flattened, tube-like structures known as cisternae, which group together to form an interconnected network. Whilst the core structure is consistent, the ER is actually divided into two regions; the Smooth ER and the Rough ER. The former is required for carbohydrate and lipid synthesis, whilst the latter looks after protein production thanks to a really nifty set of protein factories called ribosomes. In plants, the cortical ER network has been shown to play a role not only in protein trafficking but also in response to pathogens.
In this experiment, Kriechbaumer et al focused on a novel subgroup of ER proteins called reticulons. Reticulons are a family of proteins which localise at the ER, where their primary function is to promote membrane curvature by moving and shaping the ER into tubules. In Arabidopsis - a plant genus frequently used as a model for development - the reticulon protein family comprises 21 members. The attention of the team, however, was focused mainly on investigating the characteristics of just one protein, Arabidopsis reticulon 20 (RTN20). RTN20 belongs to a subgroup of reticulon proteins that possess an extended N-terminal domain. They wanted to understand more about the basic workings of RTN20 and they came to the Central Laser Facility (CLF) of the Rutherford Appleton Laboratory in Oxfordshire for answers.
In order to complete their experiment, the group excised tobacco epidermal leaf samples from infiltrated tobacco plants. They then performed FRET-FLIM using a two-photon microscope at the research complex base of the CLF to study the interaction between RTN20 and other reticulon proteins. Green fluorescent protein (GFP) and monomeric red fluorescent (mRFP) expression levels (figure 1) in the plant samples were then measured using a Nikon EC2 confocal microscope. What the team went on to discover about RTN2 – were some very interesting results.
Interpretation of the high resolution confocal microscopy data showed that RTN20, a type of N-terminal domain reticulon in Arabidopsis, had a unique punctate localisation on the ER membrane. Whilst it did indeed localise to punctae (small points) on the ER, it lacked the reticulon constriction phenotype that you would expect to observe in such a protein. The team concluded that the unique expression pattern showed by RTN20 was most likely the result of elements within its C-terminal domain or lack thereof. Not only that, but RTN20 and 19 were found to be incapable of displaying ER constriction phenotypes on overexpression, a phenomenon that would normally be seen in other previously published and recorded members of the family.
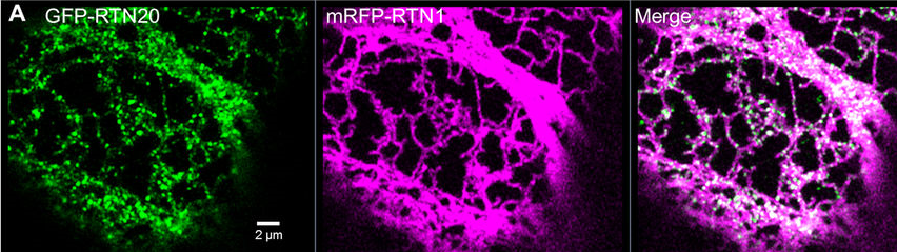
Figure 1: Pictured above are airyscan confocal images for RTN20 subcellular localisation. These images, along with others, were interpreted by the team with the help of Professor Stanley Botchway from the Central Laser Facility(credit: Kriechbaumer et al 2018).
Taking the research one step further, Kriechbaumer et al discovered that these particular reticulons may actually play a novel role in the regulation of lipids. The evidence for such a statement came from mutant studies of RTN20 and RTN19, both of which displayed a significant reduction in sterol levels in the roots. Mutant studies are particularly valuable in biology because they allow us to characterise the effect of under- or over-expression of particular genes, hence scientists can investigate the functions of particular proteins within cells. The data recorded in the paper displayed strong evidence to suppose that RTN20 does in fact play a role in sterol regulation rather than synthesis. This is because the decrease in overall sterol content in the rtn20 and rtn19 mutants could not be explained by reduction in specific sterol species.
A final conclusion made by the team was that a third homologue (3BETAHSD/D1) of these two reticulons that does not possess the typical reticulon structure was localised to ER exit sites – rather unexpectedly. This meant that the three proteins examined by the group were found in intriguingly different locations. If all of the results of this paper are collated together, it would seem that Kriechbaumer et al have presented a very strong case for considering RTN20 and even RTN19 to be a specialised subclass of reticulons involved not in ER tubulation - as is the case with other reticulons - but instead in lipid regulation. Rather excitingly, the team hope that this pioneering research will open up future avenues for investigation into the relationship between reticulon structure (C-terminal, N-terminal domains etc.) and function in plant development. Watch this space.
The research was supported by BBSRC and the full publication is available to view in Scientific Reports
For further information about the research, please contact Dr Verena Kriechbaumer (vkriechbaumer@brookes.ac.uk)
To read more news releases concerning the CLF, please visit CLF News
